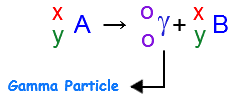
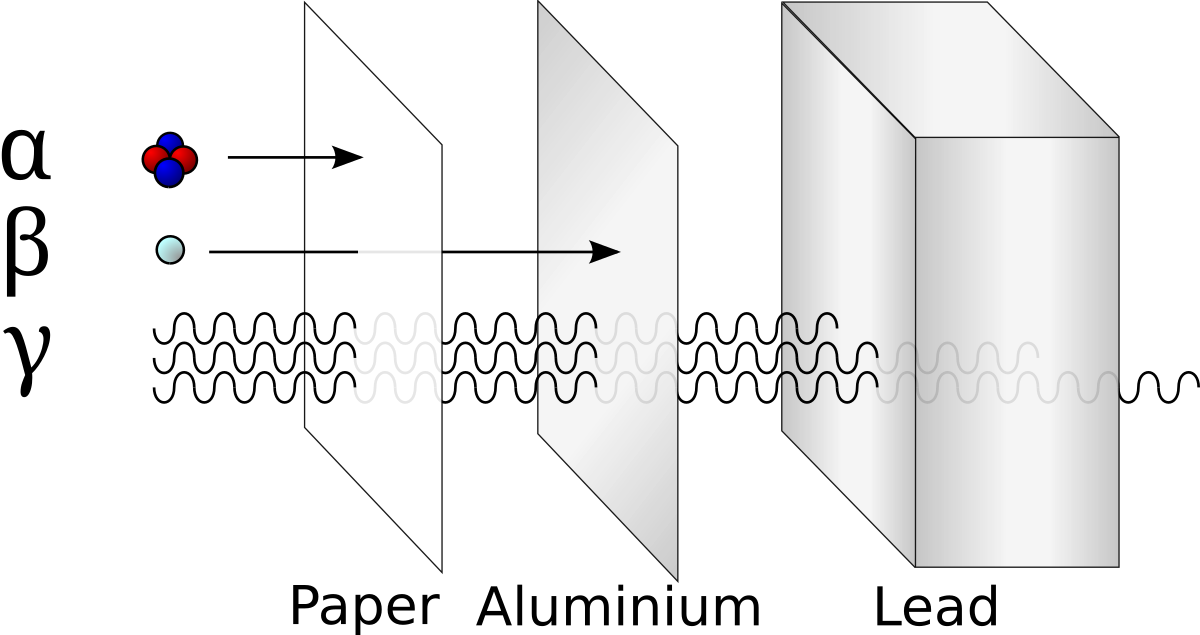
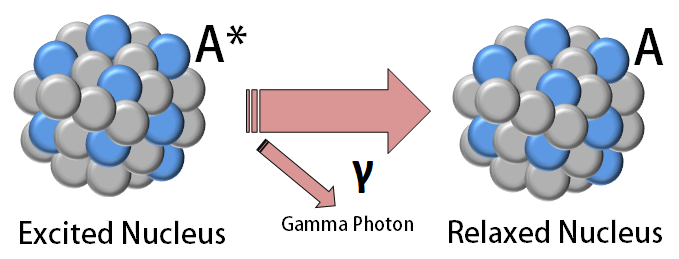
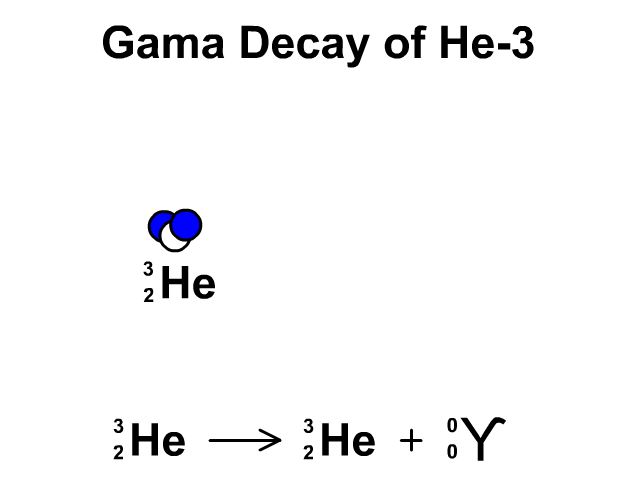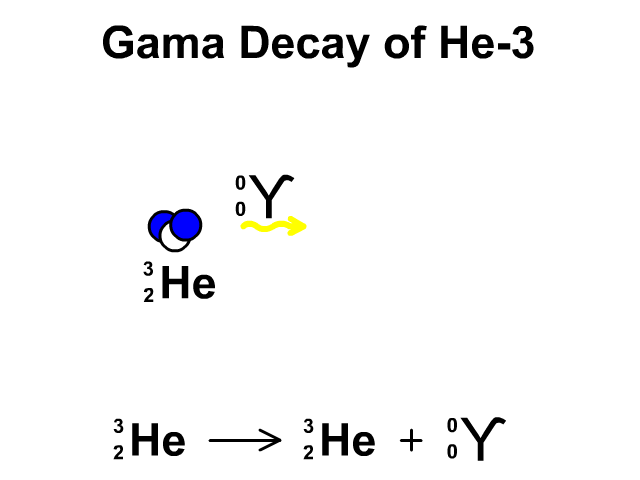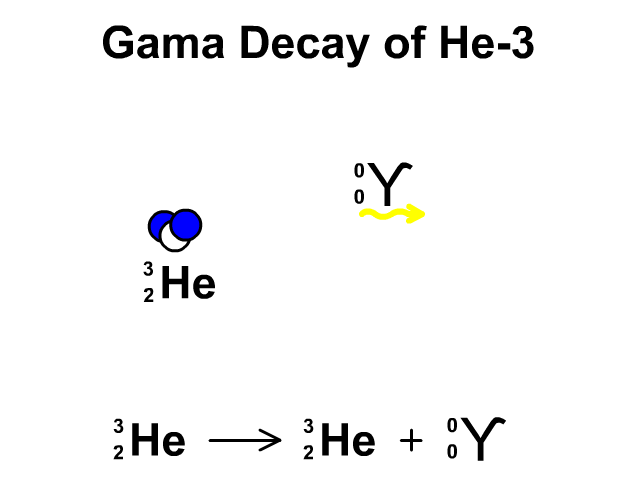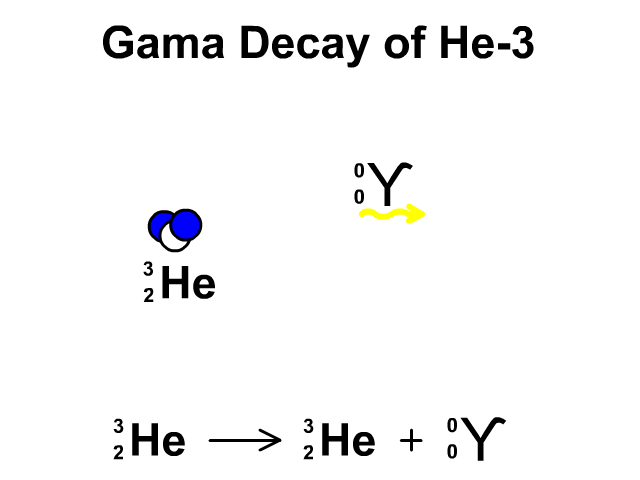
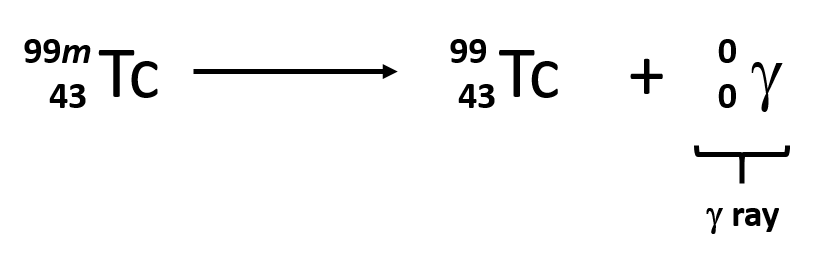
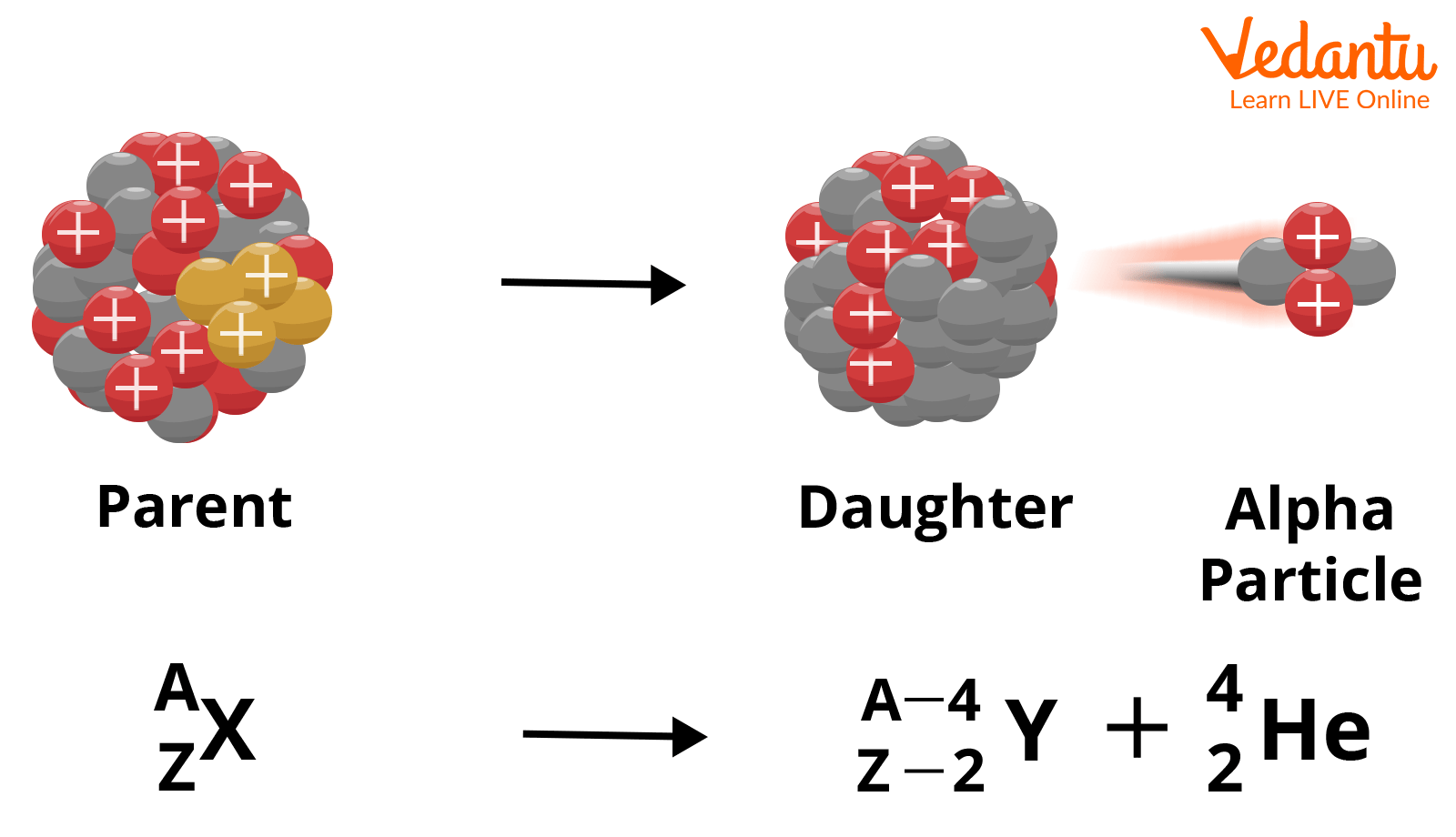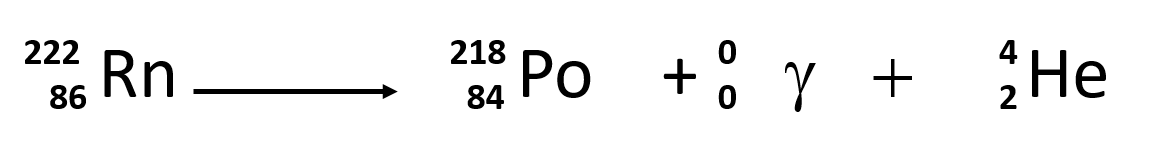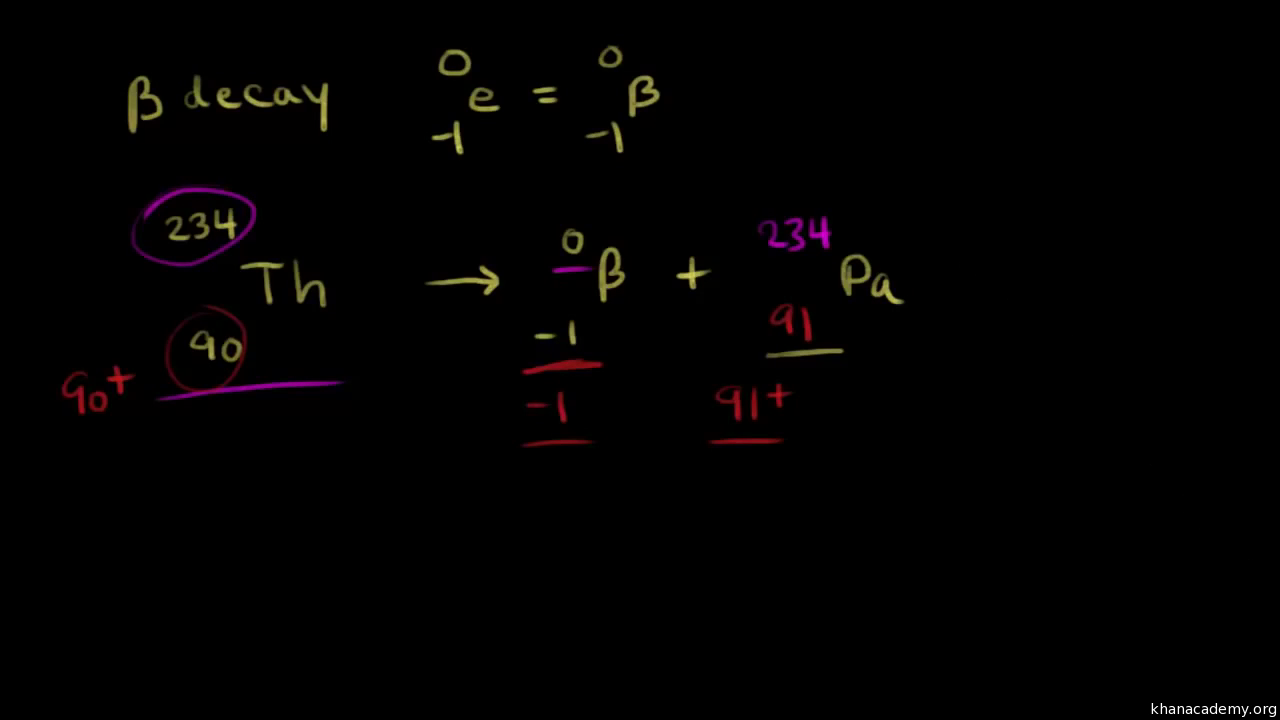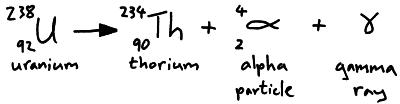Alpha Decay, Beta Decay, Gamma Decay - Electron Capture, Positron Production - Nuclear Chemistry - YouTube

Q 11 â ‹Q11 The intensity of gamma radiation from a given source is 1 On passing - Physics - Nuclei - 11928669 | Meritnation.com

Warm Up 1.What is hydronium? 2.Does it create an acid or base? 3.What is hydroxide 4.Does it create an acid or a base 5.A solution has a pH of 3. What. -
Does a single wavelength of a radiowave carry more or less energy than a single wavelength of gamma radiation? - Quora