
Stability studies needed to define the handling and transport conditions of sensitive pharmaceutical or biotechnological products. - Abstract - Europe PMC

Product Stability Testing: Developing Methods for New Biologics and Emerging MarketsBioProcess International
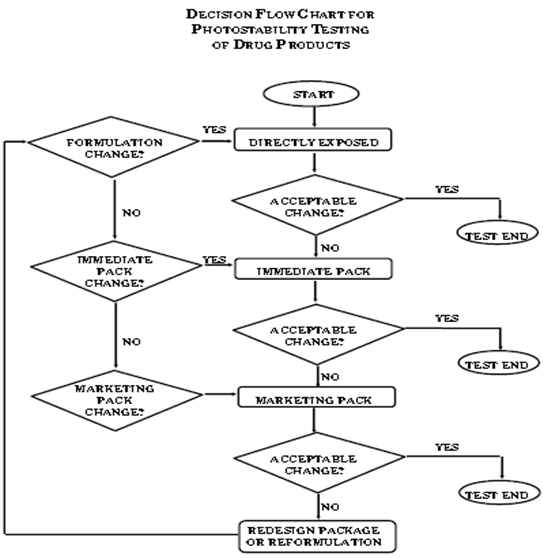








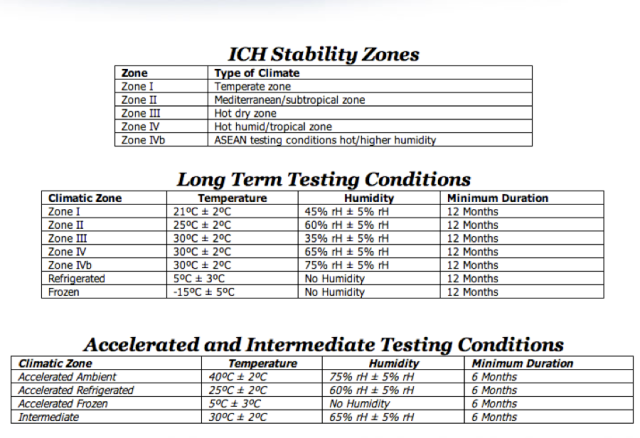






![PDF] ON STABILITY STUDIES OF PHARMACEUTICAL PRODUCTS | Semantic Scholar PDF] ON STABILITY STUDIES OF PHARMACEUTICAL PRODUCTS | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9135e2a84c016f66c9f1a94b2b8f67ae6e89e73d/2-Table2-1.png)
