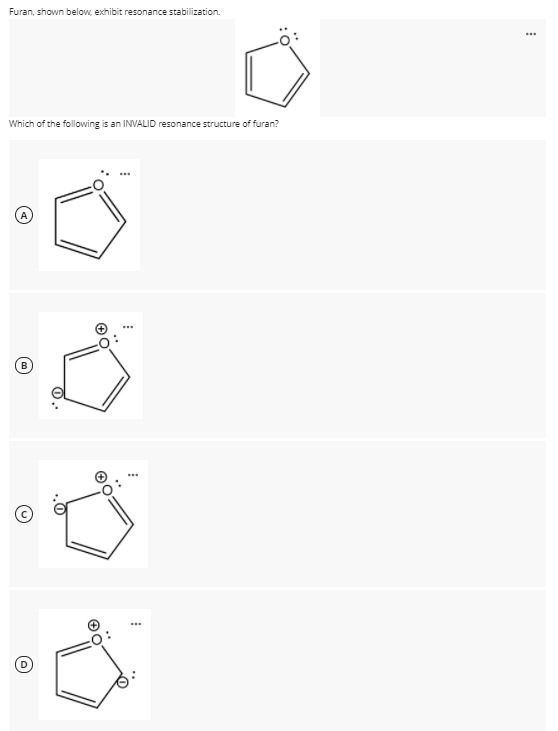
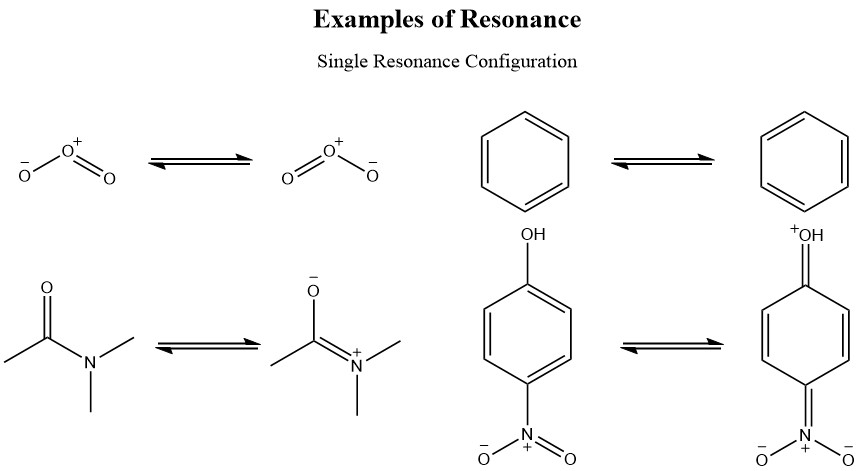
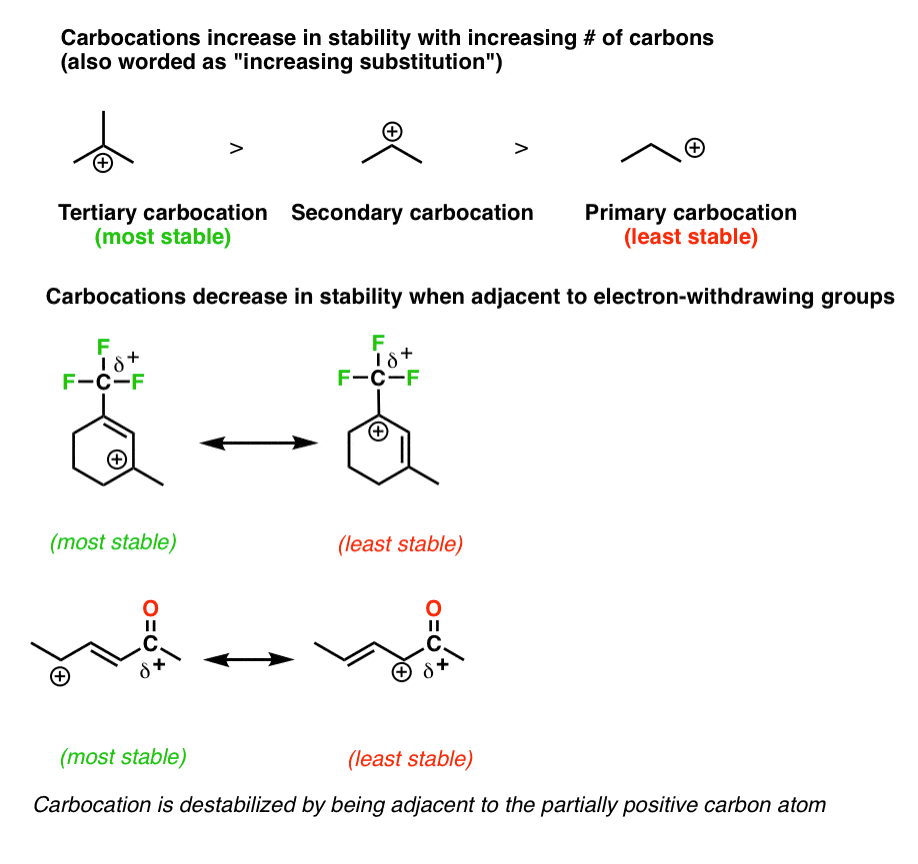
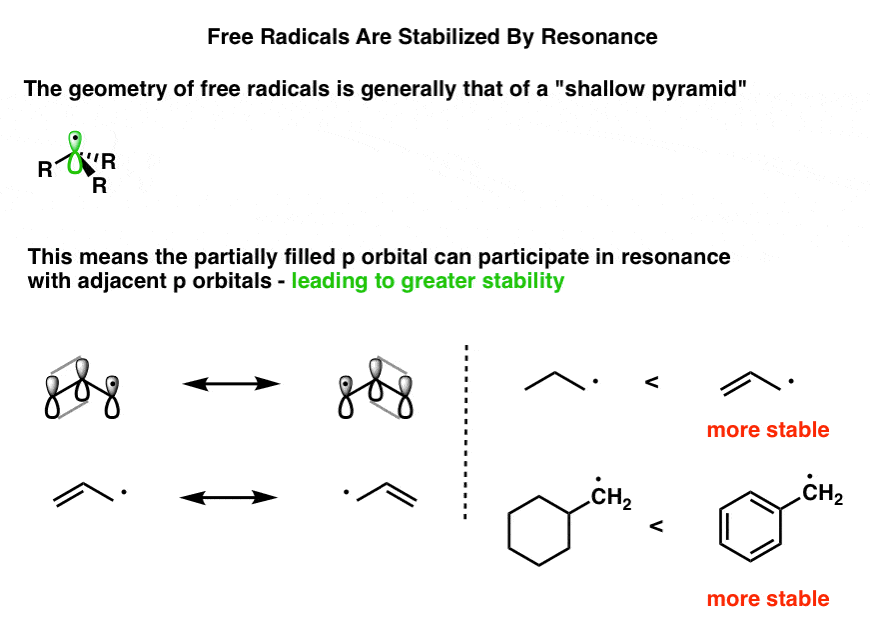
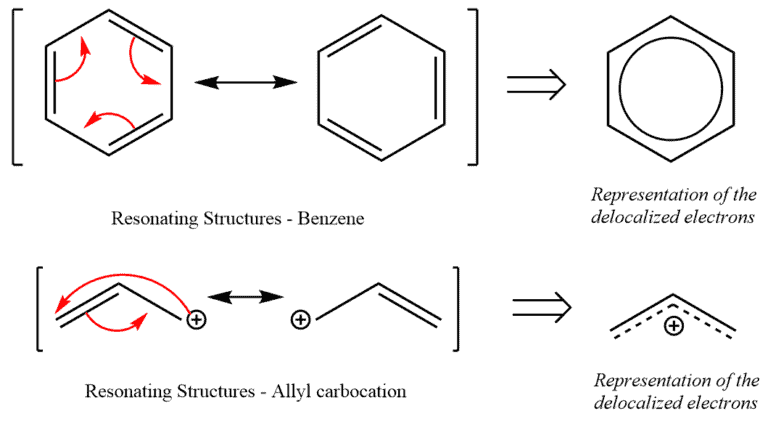
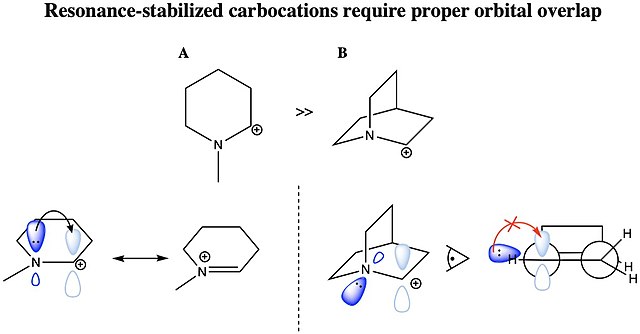
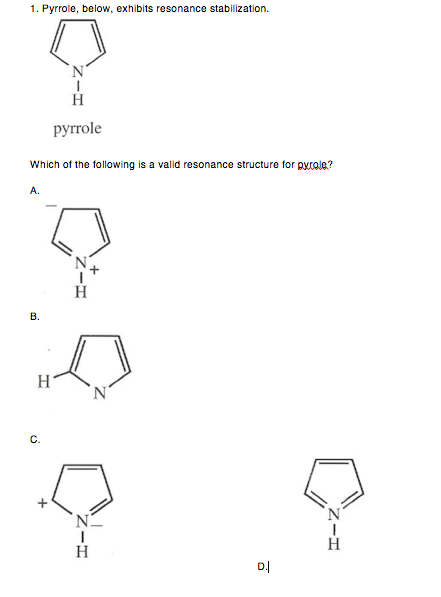
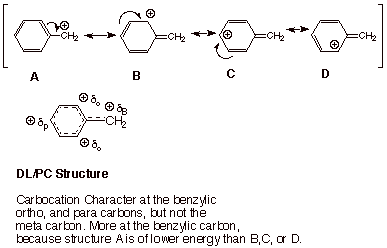
Allylic carbocations are stabilized by resonance. Draw resonance forms to account for a similar stabilization of benzylic carbocations. | Homework.Study.com

Which is more resonance stabilized between formic acid and formate ion and why? What do we mean by resonance stabilized? - Quora
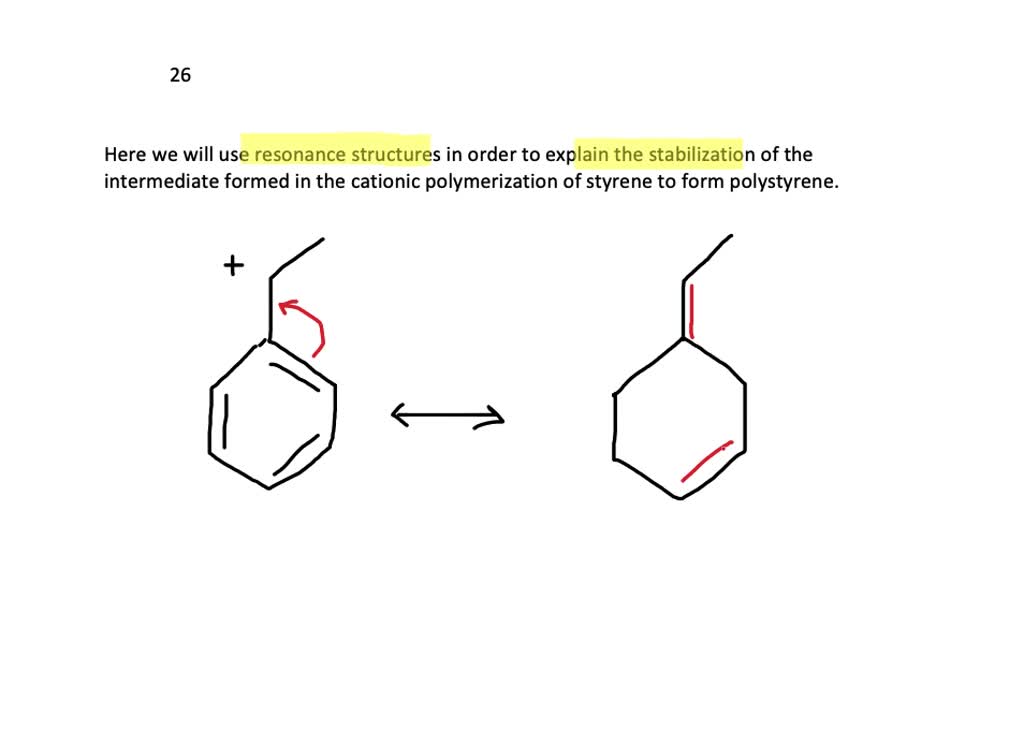
SOLVED:Use resonance structures to explain the stabilization of the intermediate formed in the cationic polymerization of styrene to form polystyrene.